Projects
Antivirals

Acyclic nucleoside phosphonates with antiparasitic activity
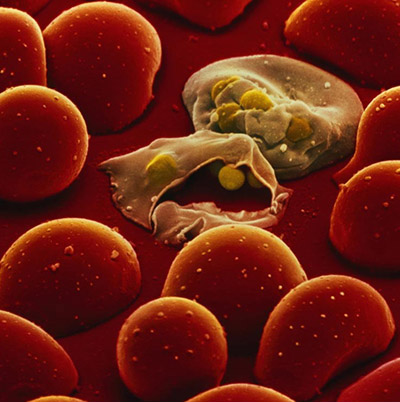
Hypoxanthine-guanine-(xanthine) phosphoribosyltransferase (HG(X)PRT) is a key enzyme crucial for the survival of the malarial parasites Plasmodium falciparum (Pf) and P. vivax (Pv). Humans possess two metabolic pathways, de novo and salvage, and hence are less reliant on the purine salvage pathway for their survival. This provides a rationale for the design of inhibitors of plasmodial HG(X)PRT, which would arrest parasitemia and, at the same time, would not be toxic to the human host. Recently, a novel group of aza-ANPs has been designed, synthesized, and studied. The low Ki values of some of these compounds for PfHGXPRT and PvHGPRT and the high selectivity in favor of these enzymes compared with human HGPRT, suggest them to be lead compounds for development as antimalarial drugs. The investigation of the inhibitory activities of ANPs is performed in collaboration with the laboratory of Prof. Luke W. Guddat, University of Queensland, Brisbane (Australia).
Immunomodulating agents
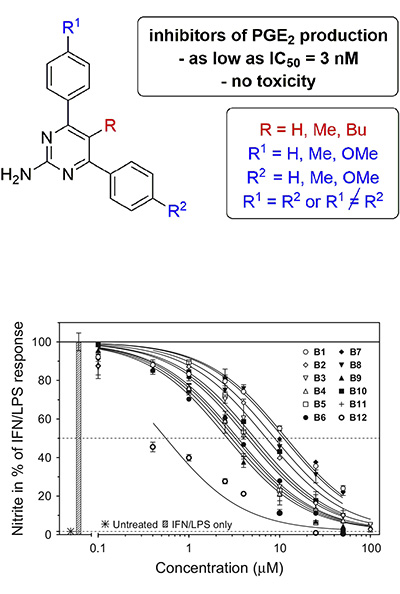
Inhibitors of adenylate cyclase