About our group
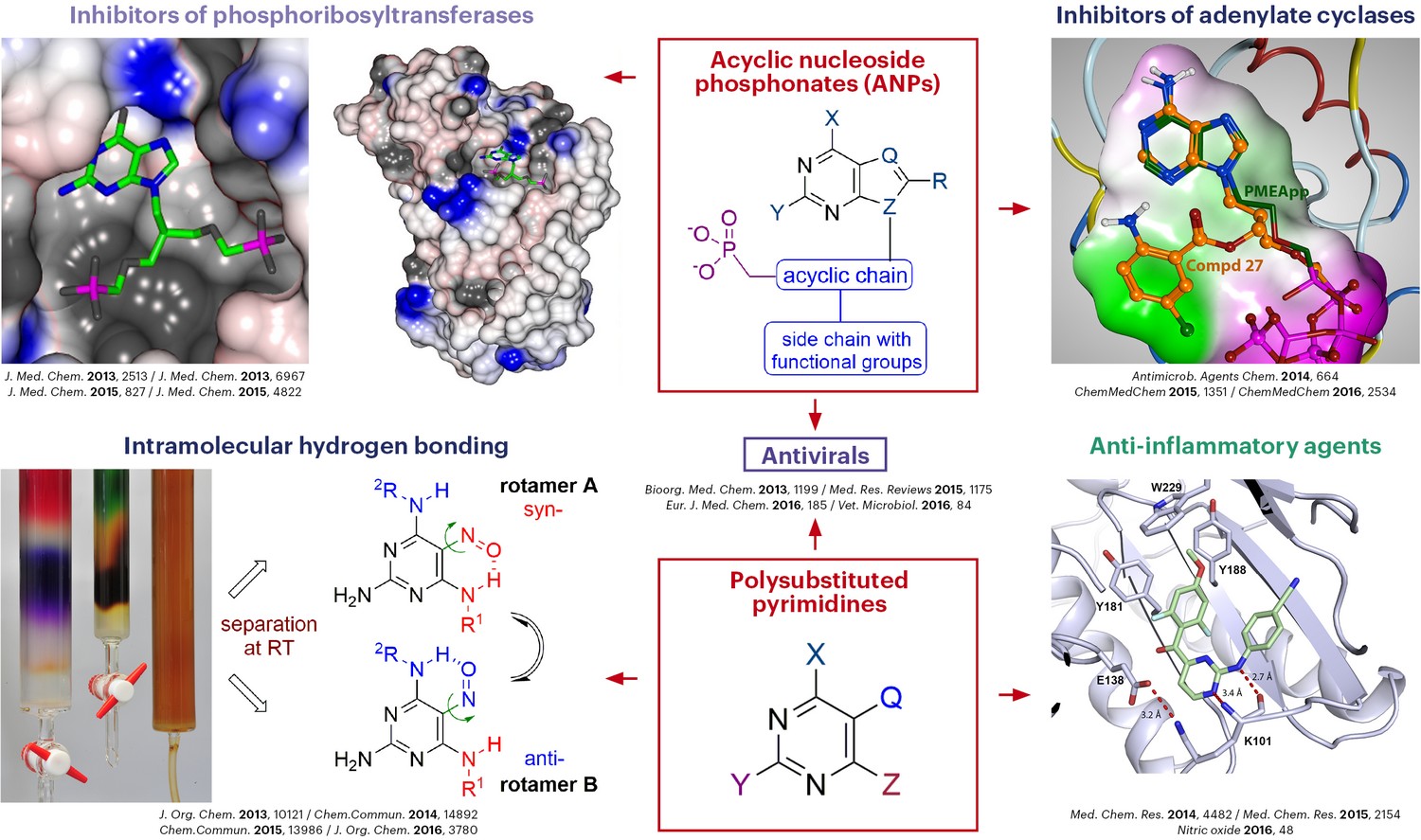
Research projects of the group deal predominantly with development of inhibitors of enzymes involved in the metabolism of nucleosides and nucleotides. Rational design of novel types of modified nucleosides and nucleotides, acyclic nucleoside phosphonates and their prodrugs in particular, is based on the previous extensive structure-activity relationship studies carried out in our group. For the syntheses of target compounds, efficient methodology is developed using diverse methods of modern organic chemistry. Target structures are designed with respect to their anticipated antiviral, antiparasitic, antibiotic, cytostatic, and anti-inflammatory properties based on inhibition of key enzymes. The mechanism of action of prepared antimetabolites and their potential biomedicinal applications are studied in collaboration with numerous scientific groups worldwide.
Publications
All publications






